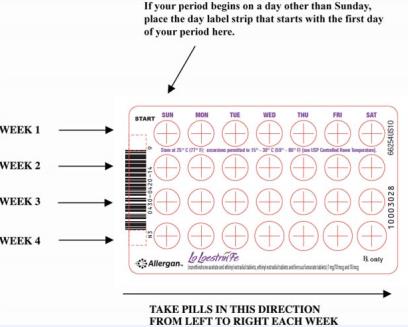
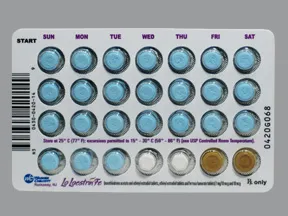
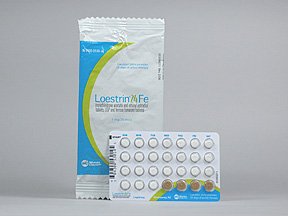
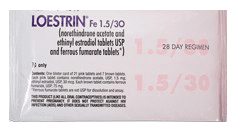
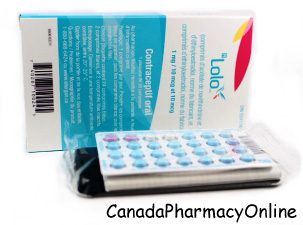
Please see full Prescribing Information, including Boxed Warning, and Patient Information for Lo Loestrin® Fe (norethindrone acetate & ethinyl estradiol tablets, ethinyl estradiol tablets & ferrous fumarate tablets): https://goo.gl/Imstbz | Please see




:max_bytes(150000):strip_icc()/VWH-EllenLindner-ExtendedCycleBirthControlPill-Standard-27fd08ef5b7e460ca1d31cc353d46b76.jpg)